Ashland biotech firm plans new clinical trials, second spin-off
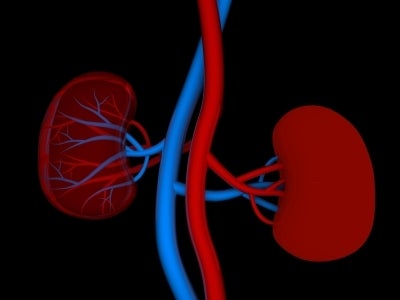
Ashland biomedical device maker BioSurfaces Inc. has kept a relatively low profile through much of its 12-year history. But new developments are creating some noteworthy buzz for the small company and its portfolio of products designed to treat renal, heart and vascular diseases.
This week, the U.S. Small Business Administration named BioSurfaces the state’s Microenterprise of the Year for its “life-changing “medical technologies, according to the SBA. Those technologies, which include artificial blood vessels, stents and sutures, employ the company’s patented electrospinning process.
BioSurfaces’ first spin-off, NuVascular Technologies, was launched last year. Its focus is bringing to market a material called NuSpun Access Graft, which was created to mimic the body’s own cell scaffolding. It’s intended for use in the company’s artificial arteries, which would be implanted in the forearms of patients in need of hemodialysis for end-stage kidney disease.
Clinical trials may begin next year
The devices allow drugs to be infused into the products’ fibers to promote healing after they’re implanted, and the materials have properties that allow the artery to seal after needle injections. This makes the products more tolerable for patients and less likely to fail than those that have been on the market for decades, according to Matthew Phaneuf, who founded the company with his wife, Tina.
Phaneuf said NuVascular hopes to complete pre-clinical trials by the end of the year, then begin clinical trials in the first quarter of 2016. After a dozen years of development, the biochemist said he and his wife, a textile chemist, are excited that commercialization is in sight. Launching NuVascular was a turning point, he said.
“It was time for us to grow up and start getting a product out that could help people,” said Phaneuf, who estimated that about 500,000 Americans require dialysis due to renal failure.
Wound care next?
Once clinical trials begin, Phaneuf said he expects it will take about two years to bring NuSpun to market (though a version of the product that doesn’t use drugs faces a shorter approval process) in the U.S. and Europe, a relatively short time period in the world of medical device approvals. He noted that the materials used in the platform are already used in different ways by other companies, so the approval process isn’t as involved.
But NuVascular is just one piece of the business strategy for BioSurfaces. The company plans to spin off multiple companies, as the NuSpun platform can be applied to many different medical devices for treatment of myriad medical conditions. The next one the Phaneufs are working on is called EmergeStat, and that company will specialize in wound care products. EmergeStat has not been officially rolled out, and probably won’t be until NuVascular products approach commercialization, Phaneuf said.
Diabetes boosts need for products
The common thread between NuVascular and its planned sister company is the population their products treat. Phaneuf said the increasing incidence of diabetes in the U.S. has led to an increase in renal and vascular disease and that population will only grow. Plus, many of these patients are now covered by health insurance after the passage of the Affordable Care Act in 2010.
“You’re going to get more patients that will get more treatment and come out of the shadows,” Phaneuf said. “So the numbers will go up.”







0 Comments